MJ14 P12 Q5 Percentage Uncertainty in Temperature Change | May/June 2014 | CAIE A Level 9702 Physics - YouTube

Improvement of reference gas calorimeter to measure the gross calorific value of the reference methane gas having high purity (99.995 %) and calculation of uncertainty
Heat Capacity Uncertainty Calculation for the Eutectic Mixture of Biphenyl/Diphenyl Ether Used as Heat Transfer Fluid: Preprint - UNT Digital Library
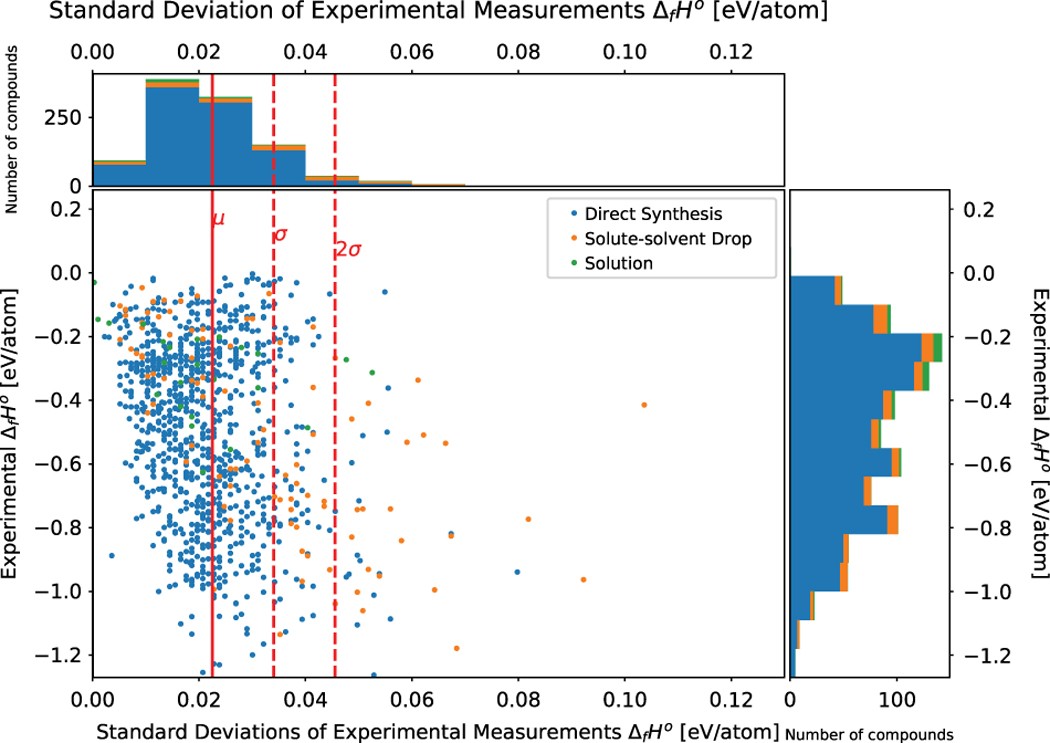
Experimental formation enthalpies for intermetallic phases and other inorganic compounds | Scientific Data

Calorimetry Problems, Thermochemistry Practice, Specific Heat Capacity, Enthalpy Fusion, Chemistry - YouTube

Towards accurate measurements of specific heat of solids by drop calorimetry up to 3000 °C - ScienceDirect

A level Experimental methods for determining enthalpy changes treatment of results bomb calorimeter sources of error graphical analysis KS5 GCE chemistry revision notes
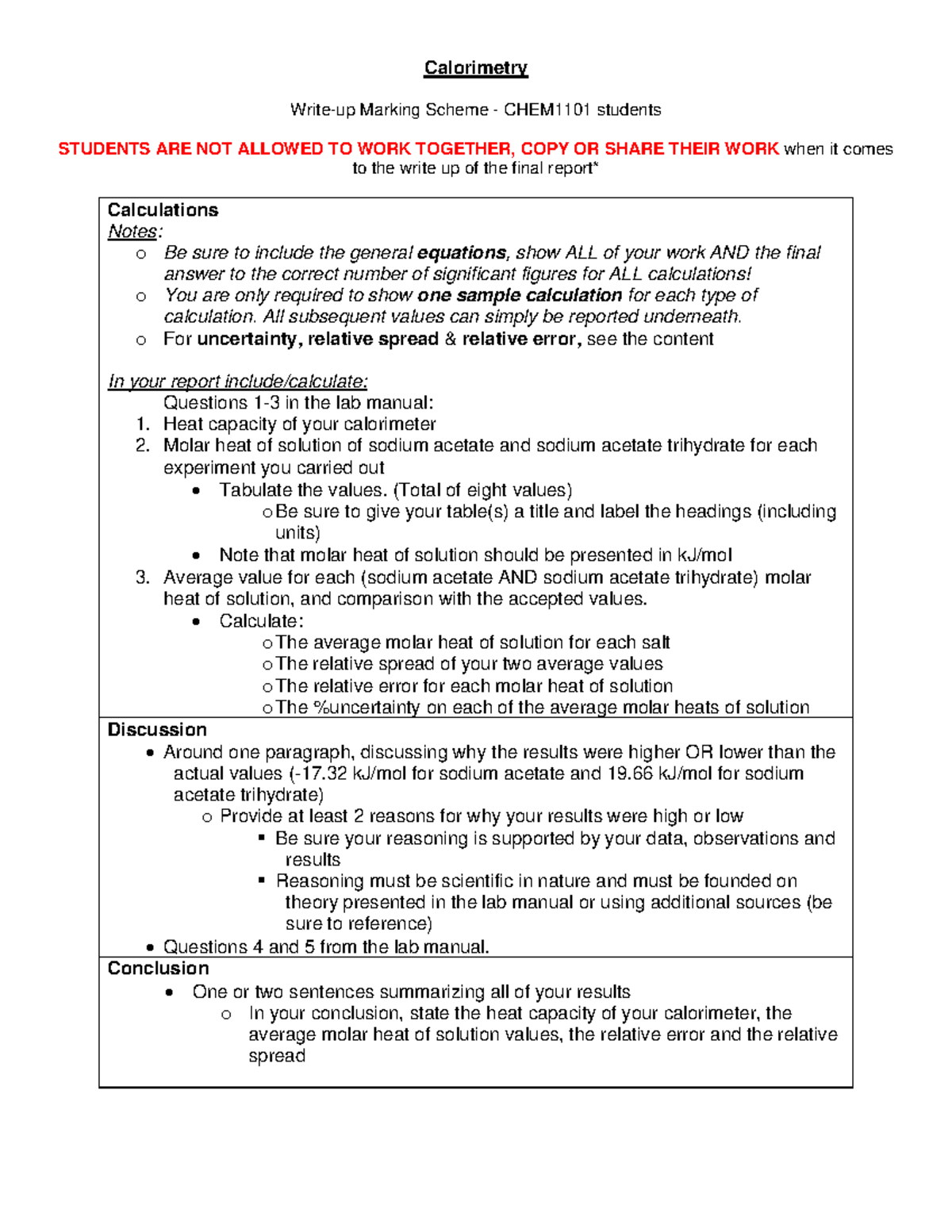
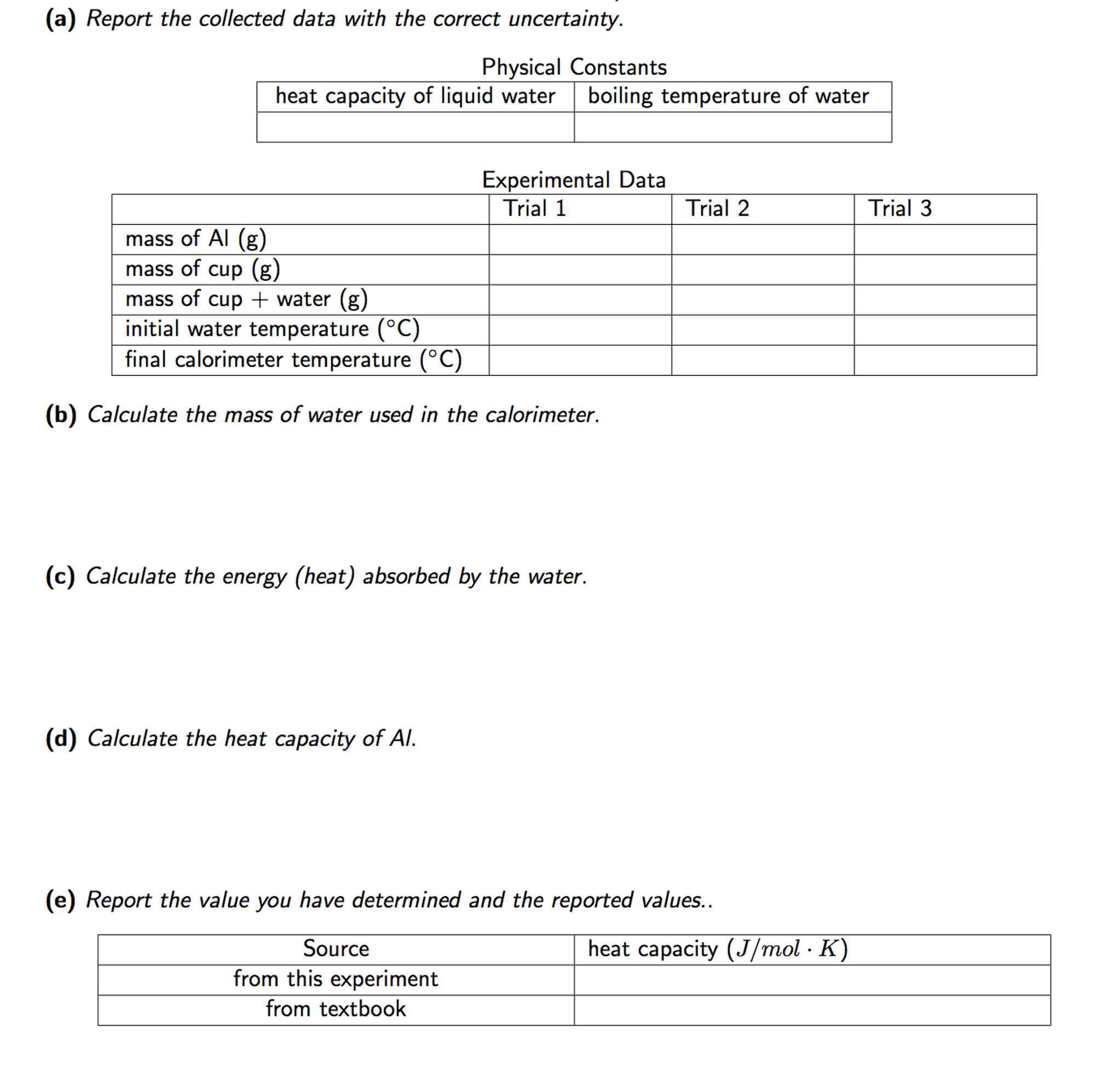
Calorimetry - lab - Calorimetry Write-up Marking Scheme - CHEM1101 students STUDENTS ARE NOT ALLOWED - Studocu
How to calculate the uncertainty of C heat capacity in the following formula knowing the measure and uncertainty to M1, M2, T1, T2 ? M1 c1 (T1- Tf) = m2 c2 (Tf -




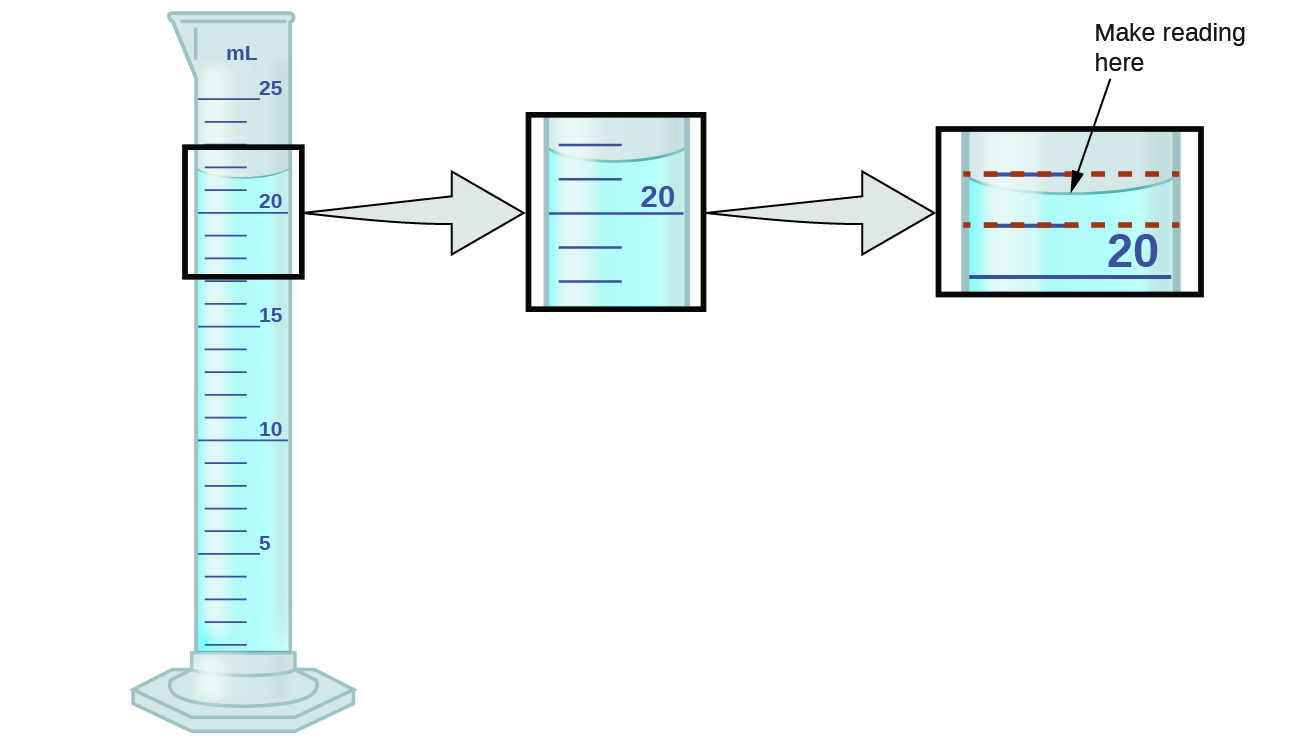

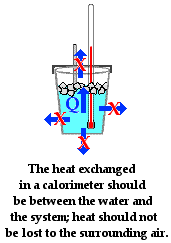




![11.1 Determine the uncertainties in results [SL IB Chemistry] - YouTube 11.1 Determine the uncertainties in results [SL IB Chemistry] - YouTube](https://i.ytimg.com/vi/B7dKWE-0FZY/maxresdefault.jpg)