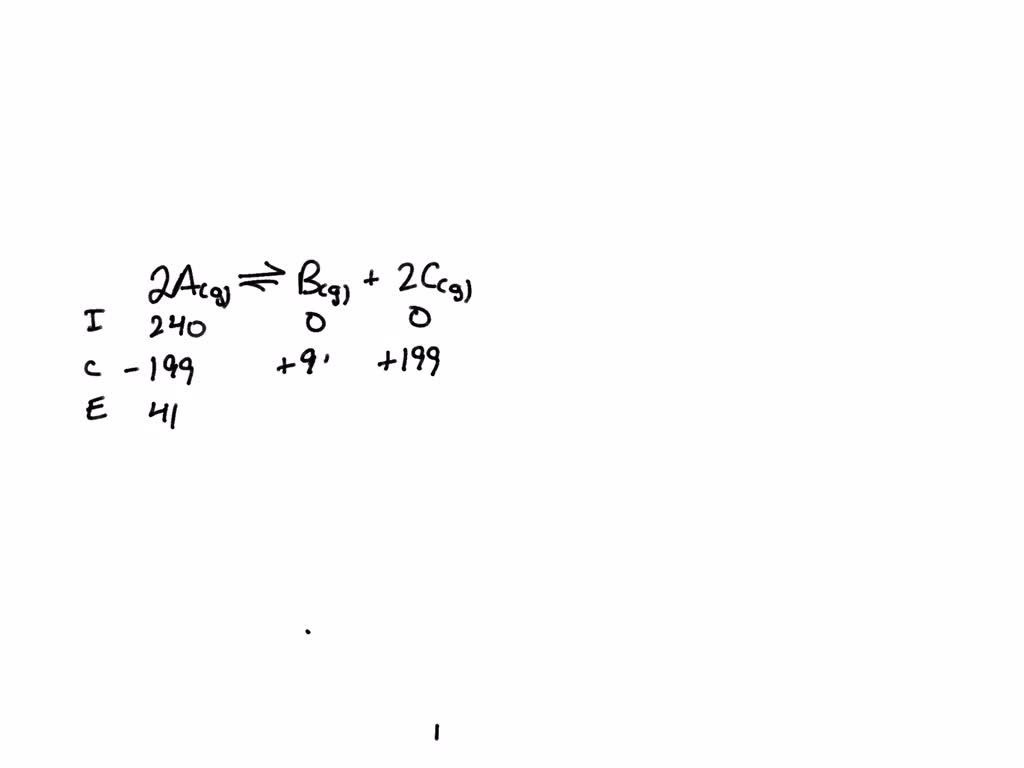
SOLVED: For the reaction 2A(g)⇌B(g)+2C(g)2A(g)⇌B(g)+2C(g), a reaction vessel initially contains only A at a pressure of PA=245 mmHg. At equilibrium, PA=41 mmHg. Calculate the value of Kp. (Assume no changes in volume

SOLVED:Ammonium chloride decomposes according to the equation NH4 Cl(s) ⇋NH3(g)+HCl(g) with Kp=5.82 ×10^-2 bar^2 at 300^∘ C . Calculate the equilibrium partial pressure of each gas and the number of grams of

amazon prime deals on beauty: Amazon Prime Day Sale 2022: Don't miss these Offers on Beauty Products - The Economic Times